 
Millikan had measured the charge of the electron qe to an accuracy of 1 percent and had raised it by a factor of 10 to a value of -1.60⋅10^-19 C within a few years. V is the voltage that holds the drop stationary. The charge of the electron may be estimated using the rearranged equation below after the mass of the drop is known. d is the distance between the plates in meters. Since we know that the voltage (V) was adjusted to balance the forces on the drop, and the electric field (E) was a product of the voltage applied, we can show it with the equation below. The mass of the drop was determined by how rapidly it descended when the voltage was turned off. This also meant that the drop was allowed to fall at its terminal velocity (v) when the voltage source was turned off. Qe is the charge of the electron in CoulombsĮ is the electric field in Newton/Coulomb 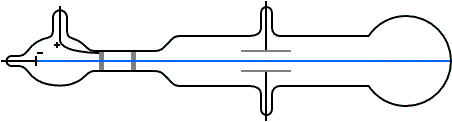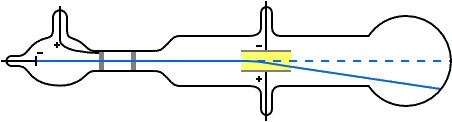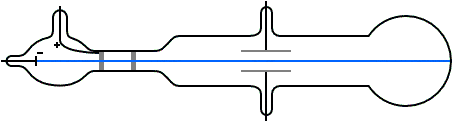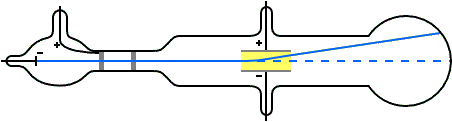
G is the gravitational constant, 9.8 m/s^2 at Earth's surface An ionising radiation source, like an X-ray tube, could also be used to charge the droplets.Ī simplified diagram of Millikan's oil-drop experiment Some were charged due to friction from the nozzle when they were sprayed. 
A potential difference was applied across the plates to create a uniform electric field. The insulator had three holes for light to enter and one for examination. In Millikan's experiment, two metal plates were stacked on top of each other with an insulating material in between. Today, it is known as Millikan's experiment. In 1909, Robert Millikan and Harvey Fletcher conducted 'the oil-drop experiment' to determine the specific charge of a single electron. These results were important, but not enough to explain the photoelectric effect. Other scientists like George FitzGerald and Walter Kaufmann also experimented with electricity and magnetism, but only found that a charge is a continuous variable. However, he could not determine the exact charge of a single electron. JJ Thomson made progress by measuring the ratio between an electron's charge and its mass (qe / me), and estimating an electron's mass to be about 5.56 ⋅ 10 ^-4 times that of a hydrogen atom. Scientists change them as new information becomes available.JJ Thomson working on his experiment with cathode rays Rather, they are the best we have at the time. We now know that even the Rutherford model was not completely correct. The Rutherford model was the best explanation of atomic structure for his time. He also agreed with Nagaoka that the electrons move in circular orbits outside the nucleus. Rutherford showed that the atom contains a small, massive, positively charged nucleus. In 1911, Rutherford showed that Thomson's model was "wrong": the distribution of positive and negative particles was not uniform. The electrons revolved around the nucleus, like the rings revolving around Saturn. 
He suggested that the atom had a massive nucleus. The Japanese physicist Hantaro Nagaoka rejected Thomson's model. Thomson's model was correct at the time, because it explained all that scientists then knew about the atom. Many North Americans would compare it to the raisins and nuts in a spherical Christmas cake. Others (not Thomson) compared it to a British dessert called plum pudding, so his model came to be known as the plum pudding model. These orbits were stabilized because, when an electron moved farther from the centre of the positive cloud, it felt a larger net inward force because caused by all the positive material inside its orbit. He assumed that the electrons were uniformly distributed and free to rotate in rings inside a sphere of positive charge. North Americans would call it a "Christmas cake" model. Thomson developed his model of the atom in 1904. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
